Model the Complexity
Black, Gray, Blue, Purple, or Green
What color is our low carbon hydrogen future?
By Stephen Ferguson
Energy is the only universal currency. Since the beginning of the Industrial Revolution, the human race has become increasingly dependent on the cheap and plentiful supply of energy. It feeds us, lights our evenings, fuels our movements, keeps us warm in the winter and cool in the summer, and provides endless piles of disposable “things” that we use as a temporary substitute for happiness.
The problem is that currently, about 87% of that energy comes from the combustion of the decomposed remains of ancient organisms (natural gas, crude oil and coal). Hydrocarbons are cheap and plentiful and are (not surprisingly) principally made from various combinations of carbon and hydrogen atoms.
During the combustion process, the carbon atoms combine with oxygen atoms in the atmosphere and produce copious amounts of CO₂, which is a greenhouse gas, which we have belatedly discovered is the principal cause of climate change. Unless we stop burning hydrocarbons pretty quickly, our species will cause permanent catastrophic damage to our planet. So why don’t we just cut out the carbon and burn pure hydrogen instead?
The good news Hydrogen is the most abundant chemical substance in the universe, constituting roughly 75% of all normal matter. Our own sun is 75% Hydrogen and 25% helium.
Hydrogen is about three times as energy-dense as natural gas (which is principally methane) and more than twice as energy-dense as gasoline. Burning hydrogen is much cleaner than burning hydrocarbons. When mixed with pure oxygen it only produces water as a by-product of combustion.
Unlike hydrocarbons, which we typically have to burn in heat engines to produce electricity, hydrogen can be used to generate electricity directly, and at much higher efficiency in fuel cells, again producing only water as a by-product. The bad news Although hydrogen is the most abundant chemical substance, hydrogen gas does not occur naturally in large quantities on Earth (although obviously, the element is present in the planet’s vast supplies of water). Hydrogen is the lightest of all elements, and any hydrogen gas that does enter the atmosphere quickly escapes the Earth’s gravity into outer space. This means that if we want to use Hydrogen as a zero-carbon fuel, we first have to manufacture it. At the moment most of the economically viable processes are far from “zero-carbon”. In 2021 so-called “Green Hydrogen” just doesn’t exist.
Most hydrogen is produced from methane and is used mainly in chemical processes
Pick a colour, any colour...
Click an icon below to find out more about Black, Gray, Blue, Purple, or Green Hydrogen
Storage and transport Our species has lots of experience in transporting liquid and gas hydrocarbons around the globe.
At the start of this article, I described how hydrogen is three times as energy-dense as methane, and twice as energy-dense as gasoline. However, this is only true per unit mass. Since hydrogen is the lightest of all elements, one kilogram of uncompressed hydrogen occupies 7.3 times more volume than a kilogram of methane. This means that a tank required to store a given amount of energy as hydrogen is much larger than for a hydrocarbon.
Another issue is that hydrogen atoms have the smallest atomic radius of any element in the universe. This means that hydrogen molecules can often migrate through the metal fabric of a storage tank, escaping into the atmosphere.
The solution to both of these problems is to compress hydrogen gas into a liquid state, in which it occupies almost 800 times less space per unit mass. However, in this state, it needs to be kept at a temperature of -241 C - which is a significant engineering challenge.
Safety Although hydrogen is not a toxic gas, it is a very flammable one. The 1937 Hindenburg disaster is etched forever in the public memory. Hydrogen has a wide range of flammable concentrations in air and much lower ignition energy than gasoline or methane
In addition, some metals can become brittle when exposed to hydrogen, so the material choice is important in the design of safe hydrogen systems.
In 2019 there were several incidents of hydrogen explosions. Firstly a storage tank at a green hydrogen production plant in Gangneung City South Korea exploded, causing two deaths and six injuries. In California, several hydrogen transporter trucks caught fire and exploded due to a leak of hydrogen. A hydrogen leak also caused an explosion in a hydrogen filling station in Norway.
Clearly, much work is needed to increase confidence in a hydrogen economy. Simcenter is used extensively in industrial gas dispersion, quantitative hazard analysis and safety risk management. Simcenter is also being used to design safe hydrogen delivery systems that can be used at vehicle refuelling stations.
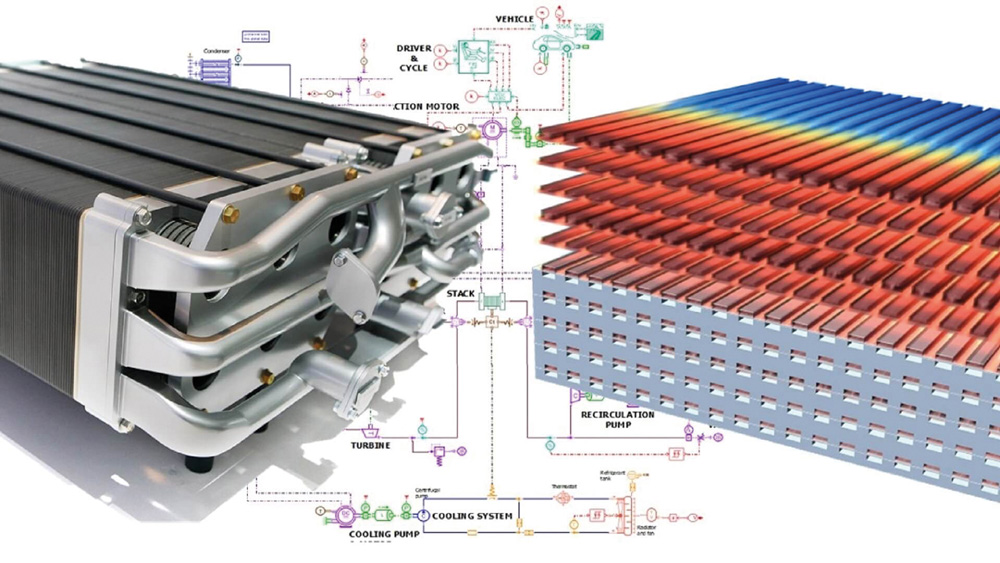
Fuel cells Hydrogen fuel cells are not new technology. Fifty years ago fuel cells provided the electricity (and some of the drinking water) that allowed the Apollo missions to reach the Moon.
Fuel cells combine oxygen and hydrogen to produce electricity. They have no moving parts, are silent in operation, and produce no harmful emissions, yielding only water as a product. They are also highly efficient, with typical efficiencies in excess of 60%, and highly scalable powering everything from laptops to power stations.
Compared with a hydrogen-powered combustion engine (described below) the fuel-to-wheels efficiency of cells is almost double. However, with this considerable promise, and decades of investment, we are yet to see the wide-scale adoption of fuel cell technology in road transportation. Part of the issue is (as described above) hydrogen has a lower volumetric energy density than gasoline. The lack of hydrogen infrastructure and safety concerns are also a problem. However, it seems certain that fuel cells will have an important role to play in our low-carbon future, and Simcenter is being extensively used to improve fuel cell technology.
Combustion engines As the world struggles to transition towards a low carbon future, it seems that the most effective method of reducing emissions is reducing combustion through electrification. This strategy obviously depends on the decarbonisation of power generation, almost two-thirds of which still depends on combustion.
There are two problems here. The first is that the power density of fuel cells and batteries is much lower than for combustion engines. This means that in transportation (which is responsible for 20% of CO₂ emissions) that it is much easier to electrify small things (cars, trains, buses, commuter aircraft) than it is to electrify large things (passenger aircraft, cargo ships, trucks). For the foreseeable future, those things will continue to be powered by internal combustion engines, powered by either biofuels or synthetic hydrogen.
Although engine manufacturers have over 100 years of experience in designing hydrocarbon fuelled engines, there is much work (and extensive simulation) required to make a new generation of reliable greener cleaner engines. Simcenter is already playing a role in that transition.
The second problem is that the rapidly growing supply of renewable electricity is intermittent. Currently, power grids depend on gas turbine generated power to provide the buffer between the “base-load” (mainly from coal or nuclear), and inherently intermittent renewable power(solar, wind, hydro). Put simply, gas turbines are what keeps the lights on when the sun isn’t shining and the wind isn’t blowing. Eventually, grid-level storage from either battery or green hydrogen might provide an alternative solution, but for now, our electricity supply is dependent on the combustion of gas.
The good news is that there is no inherent reason why gas turbines have to burn fossil fuel hydrocarbons. Gas turbines are naturally “fuel flexible”, so attention is increasingly turning towards so-called synthetic fuels such as hydrogen (H) and ammonia (NH₃). These fuels contain no carbon, and therefore do not emit any CO₂ during the combustion process.
All the world’s major gas turbine manufacturers are addressing this challenge by either retrofitting their existing turbine to work on a mixture of methane and low-carbon synthetic gases such as hydrogen and ammonia, or designing new turbines that are more naturally fuel-flexible.
Although this is an effective way of instantly reducing the CO₂ emissions of gas-fired power stations, there are significant problems to solve when using synthetic fuels. The first is the control of toxic NOₓ emissions that are a byproduct of all high-temperature combustion processes in the air (rather than pure oxygen). Although turbine manufacturers have managed to reduce these emissions to very low levels in turbines fuelled by natural gas, synthetic fuels produce different flame structures and higher temperatures. The second is that variations in flame dynamics have the potential to create thermoacoustic oscillations that could damage or limit the operation of the gas turbines.
Simcenter is being used extensively to solve both these problems and enable a new generation of cleaner, greener, gas turbines.
An (almost) hydrocarbon free hydrogen future I’ve been involved with engineering simulation for almost 30 years, and for all of that time hydrogen power has always been “just around the corner”. The reason that this has not happened so far, is that we have continued to rely on plentiful supplies of cheap hydrocarbons, and have not invested enough into solving the various technical challenges discussed in this article. However, as world governments scramble to achieve their 2030 and 2050 CO₂ emission targets, we will shortly need to confront a mostly hydrocarbon-free future. Hydrogen will play a key role in that future, and extensive engineering is the only way we will deliver it.
